
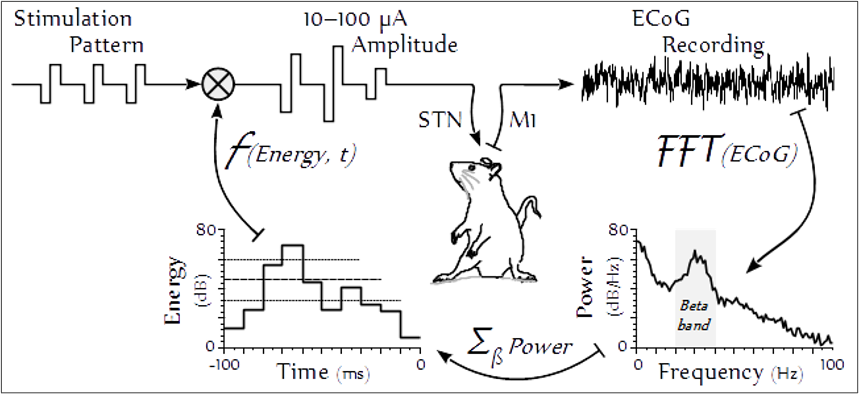
Parkinsonian Symptoms, with Deep Brain Stimulation
We aim to characterize how neurological dysfunction manifests across parkinsonian symptom domains, and how those symptoms are modulated by deep brain stimulation. While DBS is highly effective at alleviating motor deficits like tremor, its impact on non-motor and complex motor behaviors remains poorly understood. We employ behavioral assays to evaluate how both the disease state and stimulation parameters influence motivation and speech production. By quantifying these effects in parkinsonian models, we have identified critical trade-offs in therapy, such as the potential for subthalamic stimulation to dysregulate behavioral motivation or worsen hypokinetic dysarthria. Further, we investigate the specific temporal features of electrical stimuli that dictate therapeutic efficacy across symptoms. This comprehensive approach to symptom assessment ensures that our refinements to DBS technology are informed by a holistic understanding of patient quality of life.
Real-Time Bidirectional Interfaces
Traditional neuromodulation typically operates in an open-loop fashion, providing a constant drive regardless of the patient's immediate physiological state. However, bidirectional interfaces can both sense and stimulate in a dynamic, closed-loop architecture. By integrating neurophysiologic sensing with focused modulation, we are building adaptive frameworks that bridge the gap between experimental approaches and robust, clinically viable therapies. This work often leverages the Real-Time eXperiment Interface (RTXI), a high-performance software platform we helped develop to manage the rigorous timing requirements of hard real-time electrophysiological applications. Combining computational optimization with subject-specific feedback will enable responsive and highly personalized therapies in the next generation of neural interfaces.
Bicer Y, Hall J, Rampersad S, Brooks D, Dorval AD, Yarossi M (2025). “A real-time optimization-based approach to phase-specific triggering during transcranial current stimulation.” Proc IEEE Neural Engineering Research, in press.

Neuroengineering Devices
The clinical success of neuromodulation is fundamentally constrained by the resolution of the physical interface between technology and biology. Traditional electrodes often lack the spatial granularity required to selectively engage specific neural pathways without affecting adjacent structures. To address this, we are developing advanced hardware architectures, including high-density, charge-steering arrays such as the µDBS. By utilizing novel electrode geometries with thousands of individually controllable contacts, we can implement computational field shaping to accommodate surgical targeting errors and optimize the recruitment of small-diameter fibers. Our work spans the entire development pipeline—from the fabrication and bench testing of these high-resolution probes to the design of application-specific customizable architectures. These next-generation neural interfaces provide the necessary precision to shift from broad, regional stimulation toward highly targeted, patient-specific circuit modulation.
